Susceptibility
The word susceptibility comes from the Latin: susceptibilitas ‘for transferability’ and describes a physical quantity without unit, with which the magnetizability of matter within the magnetic flux density can be specified - in short, it describes the magnetic polarization in physics. It expresses itself as a material characteristic proportionality constant, which provides information about the ratio of magnetization to magnetic field strength.
In some cases it depends on other parameters such as
- Location or frequency of the magnetic field
- the previous magnetization
Susceptibility is abbreviated with the Greek letter χ. It is similar to permeability. While this refers to the whole field under the influence of a polarizing material, the susceptibility describes instead the magnetic flux and the success with which it penetrates a material.
How can magnetic susceptibility be determined?
When defining susceptibility, one should first take a look at the derivation. The most common is the magnetic volume susceptibility χ. This indicates the proportionality between the magnetization 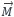 and the magnetic field strength
and the magnetic field strength  an:
an:
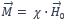 or more generally
or more generally 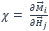 i and j are the components of spatial orientation. This takes into account the different directions in which magnetization and magnetic field can point.
i and j are the components of spatial orientation. This takes into account the different directions in which magnetization and magnetic field can point.
How can susceptibility help classify magnetic materials?
Every known substance reacts to a certain extent to an external magnetic field. For this reason, substances without a magnetic order are usually distinguished based on the susceptibility between paramagnetism and diamagnetism:
- Diamagnets (
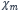 < 0): According to Lenz's rule, diamagnetic substances want to displace their internal magnetic field so that they magnetize in the direction of an external magnetic field (negative susceptibility). They are independent of temperature. Superconductors are an exception (
< 0): According to Lenz's rule, diamagnetic substances want to displace their internal magnetic field so that they magnetize in the direction of an external magnetic field (negative susceptibility). They are independent of temperature. Superconductors are an exception (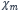 = -1). Examples of diamagnets would be hydrogen, noble gases, copper and lead.
= -1). Examples of diamagnets would be hydrogen, noble gases, copper and lead. - Paramagnets (
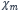 >0): In the case of paramagnetic substances, the magnetic moments align themselves in external magnetic fields and thus strengthen their internal magnetic field. Magnetization and susceptibility are positive. The temperature dependency is calculated using the magnetic susceptibility (also called the Curie constant). Examples of paramagnets are aluminum, sodium and oxygen.
>0): In the case of paramagnetic substances, the magnetic moments align themselves in external magnetic fields and thus strengthen their internal magnetic field. Magnetization and susceptibility are positive. The temperature dependency is calculated using the magnetic susceptibility (also called the Curie constant). Examples of paramagnets are aluminum, sodium and oxygen.
- Ferromagnetism: The magnetic moments of ferromagnets are aligned in a strongly amplifying manner parallel to the external magnetic field. They are completely magnetizable. Examples of ferromagnets are α-iron, cobalt and nickel.
- Ferrimagnetism: The susceptibility depends on the previous magnetization. The anti-parallel alignment of their magnetic moments ensures a different amount and thus spontaneous magnetization. Examples of ferrimagnets are magnetite (
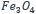 ).
). - Antiferromagnetism: Antiferromagnets are magnetically anisotropic, i.e. their susceptibility is related to the orientation of the solid in the magnetic field. If this lies in the same plane as the magnetic moments, the susceptibility and temperature are approximately linear. With vertical alignment, it is temperature-dependent. Examples of antiferromagnets are manganese and chromium.
Ferrimagnets and ferromagnets can be used as permanent magnets, since after the external magnetic field has been switched off, residual magnetization remains. Soft magnets have a susceptibility that approximates permeability. For this reason, they can be remagnetized without problems. They are ideally suited for use in generators and transformers.
 Thousands of products in stock
Thousands of products in stock